After a tumultuous 2020, we start the New Year with a modern miracle in medicines and hope – COVID19 vaccines are approved and administered in the US and around the world. I am writing this updated blog article the week of January 6th, 2021, as an update to my previous article in December 2020. The science and news advances daily; this article shares the latest research published and available through January 5, 2020.
If you have a strong anti-vaccine point of view, this article is not for you.
While receiving the COVID19 vaccine is a personal choice…you are also choosing between vaccination or getting the COVID19 infection.
Note, I am a board-certified physician who believes in science. (Neurology and Integrative Medicine). As a physician who consults and speaks on corporate wellness, I receive frequent inquiries about my thoughts on the COVID19 vaccines. I wanted to take the time to answer additional questions we have been receiving online and during our wellness workshops. At the bottom of this article, you will find the scientific resources I read to verify facts, data, and the answers to your questions.
The US Food and Drug Administration on December 18 issued an emergency use authorization for the Moderna COVID19 vaccine.
The additional and detailed medical study discussing scientific research for this vaccine is cited from the New England Journal of Medicine article in December 2020.
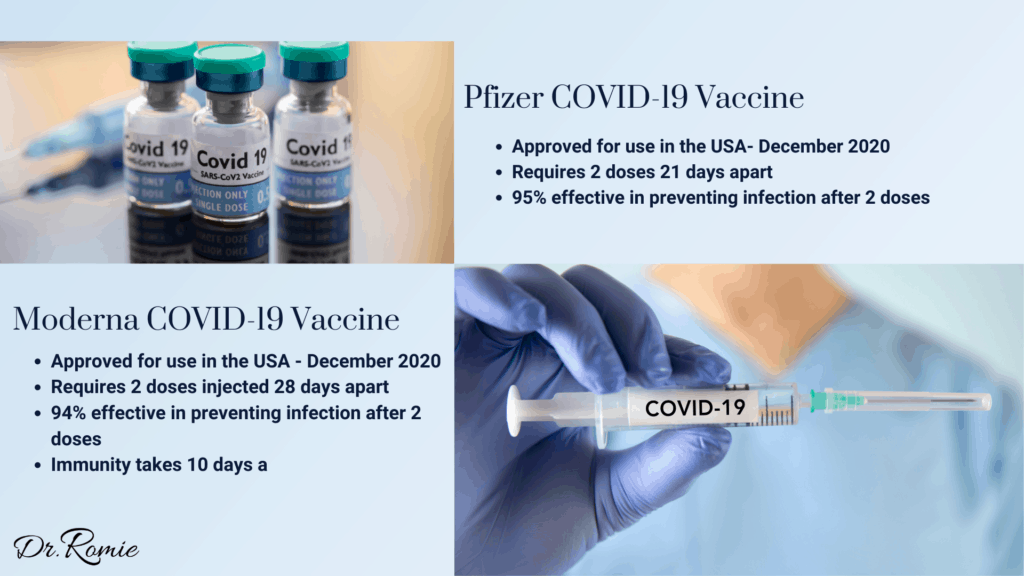
The Moderna COVID19 vaccine’s approval is now the second one available in the US after the Pfizer vaccine’s FDA approval in early December.
How Effective is the New Moderna Vaccine?
According to data presented to the FDA, the Moderna vaccine was 94.1% effective at preventing symptomatic cases of COVID19.
The clinical trial to test the Moderna vaccine enrolled 30,420 volunteers. Volunteers either received the vaccine or a placebo shot (participants did not know which one they received – known as a double-blinded placebo-controlled trial). There were 15,210 participants assigned to each group.
Why do I have to Take Two Doses of the Vaccine?
In clinical studies of both of the approved vaccines, a single dose did not induce a strong antibody response. Both vaccinations require you to receive a second so that your antibody response compares to the antibody responses that are present in people who had survived the infection.
Both vaccines require two doses. For those receiving the Pfizer vaccine, the second dose is administered 21 days after the first. Moderna’s second dose is administered 28 days after the first.
How Long Does it Take to be Protected From COVID19 Once You Get the Vaccine?
Both the Moderna vaccine and the Pfizer-BioNTech vaccine begin to protect recipients approximately 10 days after the first dose, with maximum protection after the second dose. To reach the 94-95% effectiveness, you must wait until after the second dose.
It typically takes a few weeks for the body to build immunity after vaccination. That means it’s possible a person could be infected with the virus that causes COVID-19 just before or just after vaccination and get sick. This is because the vaccine has not had enough time to provide protection.
What are the Risks of the Moderna COVID19 Vaccine?

Side effects that have been reported with the Moderna COVID-19 Vaccine include:
- Injection site reactions: pain, tenderness and swelling of the lymph nodes in the same arm of the injection, swelling (hardness), and redness
- General side effects: fatigue, headache, muscle pain, joint pain, chills, nausea and vomiting, and fever
The difference is that the Moderna vaccine’s side effects were found to be more common after the second dose. Either way, health experts said people might want to consider taking the day off work after getting the second shot.
Why Does the News Keep Changing About the COVID19 Disease, Treatments, and Vaccine?
One year ago, the global medical and scientific community had not yet heard about COVID19. Because this is a new disease process, we learn about the symptoms, long term effects, potential treatments, and prevention in real-time. Medical researchers, physicians, and scientists are sharing data globally daily, meaning the news will update at a rapid pace.
Can I Trust a Vaccine Made So Quickly?
The COVID19 vaccine is an incredible feat of modern medicine, good news. However, people may be hesitant to get it due to the creation speed, even people who vaccinate against all other diseases. This week, Gallup released a poll that now up to 63% of Americans are willing to try the vaccine, increased from 51% earlier this summer.
The mRNA vaccines are not new. The research dates back to the early 1990s, being tested for rabies, Zika virus, influenza, and other viral diseases. However, this is the first time an mRNA vaccine will be used in public.
At the time I am writing this article, we are waiting for more details on side effects in clinical trials. (As this becomes available, we will update this post). We will learn more about the side-effects as the first sets of doses are administered in the US and around the world.
Prior to being tested for viruses, mRNA technology was tested in cancer to target tumor proteins.
In January 2020, Chinese scientists released the data on the genetic code of the Coronavirus producing COVID19, and scientists around the world started to work on vaccines.
Will I be Getting the Vaccine? Absolutely.
When it is my turn, I will take the vaccine.
Given the shortage of vaccination doses, essential workers and the elderly are receiving priority as per the CDC guidelines. I am not a physician at the front lines, so I want my colleagues, nurses, and other healthcare workers who interface with COVID19 patients daily in hospitals to receive the vaccine first.
For the majority of American adults, vaccines will be available later in 2021.
What Does MRNA Vaccine Technology Mean? Will the Vaccine Give Me COVID19? Will MRNA Change My DNA?

Let’s start with how this vaccine works. This is an mRNA vaccine, which is a new technology compared to other vaccines you may have taken in the past. Past vaccines typically use a live but weakened “attenuated” virus, meaning it contains dead viral material (“inactivated” virus), or a piece of the virus’s protein, or even a toxin produced by the virus.
The Covid vaccine is very different. It contains mRNA (messenger genetic material) that encodes for the Covid spike protein. The mRNA vaccines teach cells how to make a protein—or even just a piece of a protein—that triggers an immune response inside our bodies. That immune response, which produces antibodies, protects us from getting infected if the real virus enters our bodies. The mRNA helps our cells create a spike protein.
When your cells release just the spike protein, it will stimulate your immune system to form antibodies to the COVID19 spike protein without you getting sick. There is no possibility of getting Covid from the vaccine. When your body is subsequently exposed to COVID19, it will quickly recognize the spike protein and destroy it before it can make you sick. This was 95% effective in preventing COVID19, which is an even better percentage than most other vaccines. However, you must take both doses of the Pfizer vaccine (about 3-4 weeks apart).
(In contrast, when you are sick with Covid, the virus hijacks your cell to produce many copies of the entire virus. The virus destroys the cell, busting it open to release its newly formed viral particles.)
Did the Pharmaceutical Companies, Government, or FDA Cut Corners to get the Vaccine Produced so Quickly?
Because the virus’ gene sequence was available in January, the vaccine was developed from the technology we had from the prior mRNA vaccine research on other Coronaviruses and cancer research in March. Usually, there would be several months for the FDA to evaluate the research prior to approving Phase 1 trials. However, given the pandemic’s urgent nature, the review was done quickly, which cut out months of waiting, but no one cut any corners.
Will There be Other Vaccines Available in the US?
As of this blog article’s published date, the United States FDA has approved two COVID19 vaccines by Pfizer and Moderna.
According to the CDC, there are additional vaccines in phase 3 clinical trials in the United States. In this stage of a clinical trial, a large number of volunteers receive the vaccine to research whether it is effective and to document side effects.
As of December 28, 2020, large-scale (Phase 3) clinical trials are in progress or being planned for three COVID-19 vaccines in the United States:
- AstraZeneca’s COVID-19 vaccine
- Janssen’s COVID-19 vaccine
- Novavax’s COVID-19 vaccine
Resources:
- Moderna Vaccine information from FDA website: https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-covid-19/moderna-covid-19-vaccine
- Facts about the Moderna Vaccine (EUA from FDA): https://www.fda.gov/media/144638/download
- Center for Disease Control (CDC) information on Covid19 vaccines: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/index.html
- Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine: https://www.nejm.org/doi/full/10.1056/NEJMoa2035389
- Moderna’s COVID19 Vaccine proven effective in NEJM: https://www.medscape.com/viewarticle/943467
- FDA authorizes the use of Pfizer COVID19 vaccine: https://www.medscape.com/viewarticle/942479
- New England Journal of Medicine editorial on “A New Vaccine to Battle COVID19” https://www.nejm.org/doi/full/10.1056/NEJMe2035557
- How COVID Vaccines were developed so quickly: https://www.medscape.com/viewarticle/941719
- COVID 19 Practice Guidelines: https://emedicine.medscape.com/article/2500114-overview
- mRNA vaccines a new era in vaccinology: https://www.nature.com/articles/nrd.2017.243
- Vaccines facts on WebMD: https://www.webmd.com/vaccines/covid-19-vaccine/news/20201216/covid-19-vaccines-101-faq
